“Now everything is in one system, so you have a perfect overview.”

Iris Scheepers
Senior Manager, QM Systems and Audits,
BASF


Ensure access, visibility, and control with automated document review and approval workflows.
Veeva QualityDocs is an end-to-end content management solution for both regulated and non-regulated quality content. Veeva’s proprietary GxP content reference model enables best practices and industry standardization. The application manages content throughout its entire lifecycle, including embedded Office365 connectivity, workflow automation, and deep and efficient search capabilities. It allows internal and external parties to collaborate and share information, such as procedures, policies, work instructions, quality agreements, and batch-related documentation, in a controlled manner directly within the system.
Announced 2013 | Status Very Mature | Customers 100+
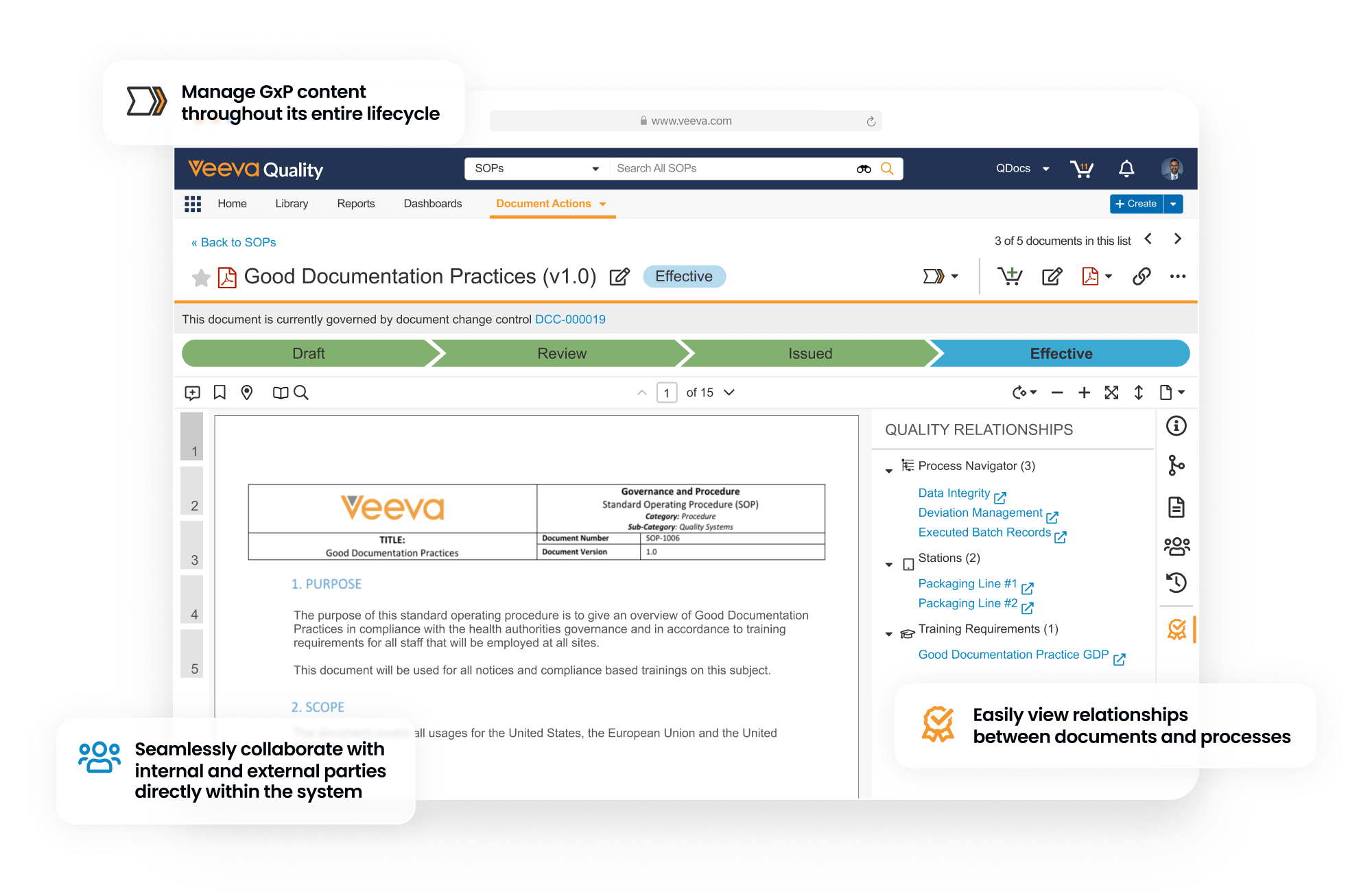
Establishing a Single Source of Truth for Document Management
Generate translations of documents such as SOPs into various languages to improve overall cycle times.
Demonstrate compliance with tracked reviews and approvals and automated workflows across the content lifecycle.
Intuitive, easy-to-use user interface drives adoption and efficiency.
Detailed audit trails and powerful search facilitate better audits and inspections.







Senior Manager, QM Systems and Audits,
BASF


Global Head Corporate QM System,
PMI
.png?width=1830&height=1068&name=Ren%C3%A9%20Bohren%2c%20Global%20Head%20Corporate%20Quality%20Management%20System%20at%20Philip%20Morris%20International%20(PMI).png)
Veeva QualityOne helps manufacturers deliver high-quality products that consumers trust. The cloud-based platform unifies applications, processes, and partners across content management, Quality Management System (QMS), food safety management, and training.
Follow Us on Social
