“Veeva LIMS can significantly streamline lab operations by consolidating the numerous systems, paper processes, and spreadsheets currently used to manage QC into one application.”
Mike Jovanis, President, Quality Cloud
Veeva

Increase lab capacity, improve GMP compliance, and accelerate batch release.
Veeva LIMS optimizes batch release testing and stability study management for the quality control lab. It drives detailed sample management, digital test method execution, specification adherence, and review by exception to accelerate the release of product.
LIMS promotes compliance by verifying user qualifications from Training, displaying effective test method procedures from QualityDocs, and initiating lab investigations directly in QMS from out-of-specification (OOS) results.
Announced 2021 | Status Early | Customers 11–50
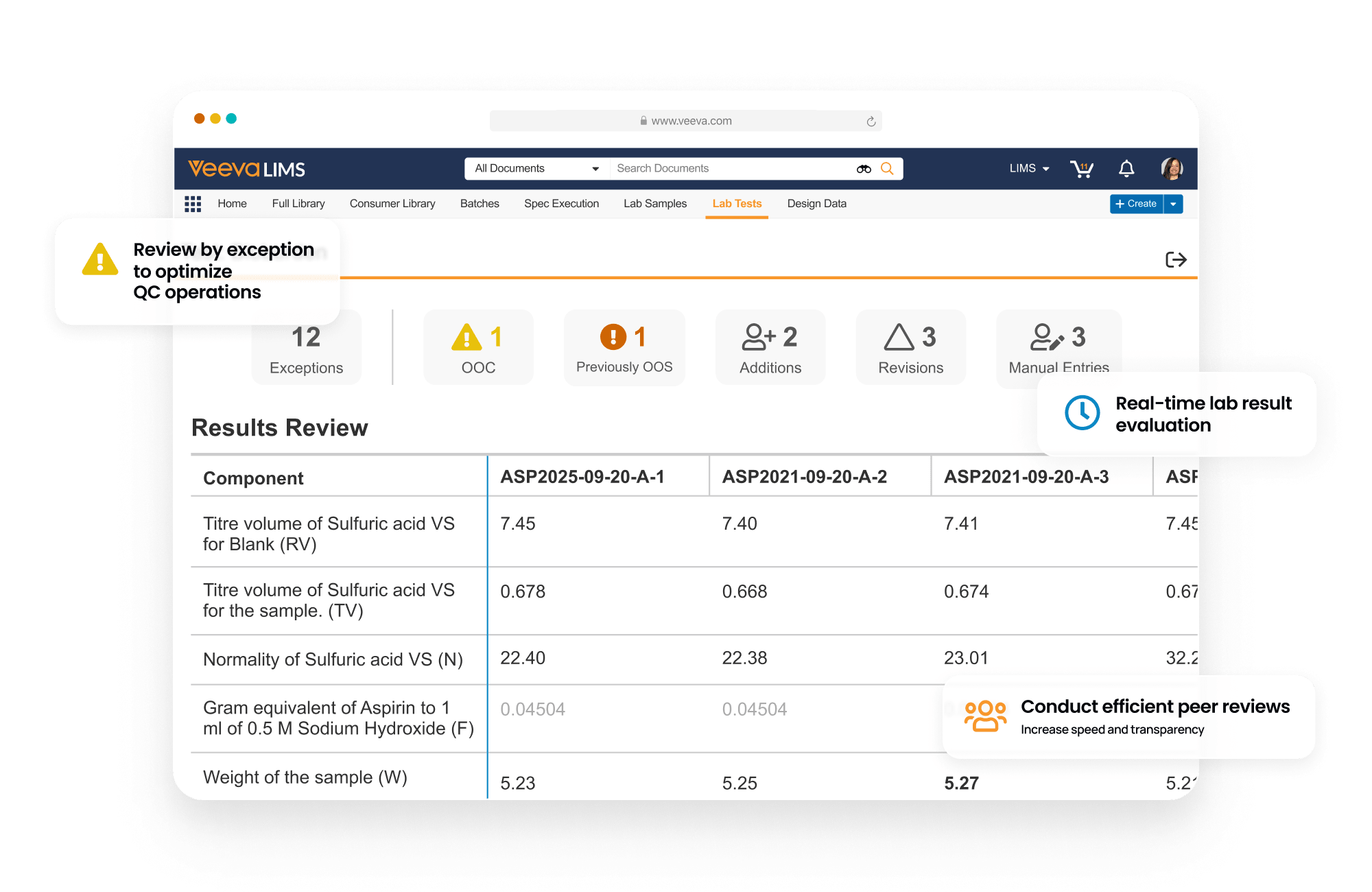
See how Veeva LIMS can modernize your QC operations
Replace legacy systems and paper processes to increase reliability and accuracy with one unified solution.
Streamline end-to-end QC data management and test execution
processes to break down silos across systems, teams, and data.
Reduce errors and improve speed with seamless quality processes and
automated workflows.
Veeva
Veeva QualityOne helps manufacturers deliver high-quality products that consumers trust. The cloud-based platform unifies applications, processes, and partners across content management, Quality Management System (QMS), food safety management, and training.
Follow Us on Social
